Hydrostatic manometers
In this kind of manometer, the difference between two pressures is balanced by the difference of level in a liquid. To measure pressure in a liquid, a manometer can use the working fluid itself, this is obviously not the case when pressure is measured in a gas: the choice of the manometer fluid (or separating liquid) will be affected by the pressure field, the required sensitivity and the physical and chemical properties of the liquid. The properties of the most common manometer liquids are shown in
Table 1.1.
The liquids that work best are those that are chemically stable, are not affected by moisture and have the following qualities: low viscosity, as this reduces the response time; low surface tension, low volumetric expansion coefficient, low volatility, consistency, and resistance to contamination.
1.2.1 The U-tube manometer
The fluid is contained in a glass tube with U-shaped vertical arms (Figure 1.1) with internal diameters more or less constant. The pressures pj and p2 will be applied through suitable piping connection to the two arms of the manometer. If the difference between the heights of the columns of fluid in the two arms is Ah, it will be:
Л – p = p – Pa)gAh = PgAh (1.1)
where g is the acceleration of gravity and pt and pa are the densities of the manometer liquid and of air, respectively. The approximation made when
|
Liquid |
Density at 15°C (g/ml) |
Surface tension (dynes/cm) |
Angle of contact |
Coefficient of cubical expansion per С x 105 |
Remarks |
|
Water |
0.999 |
74 |
8°(*) |
5°C – 10°C 5.3 10°C – 20°С 15.0 |
(*) 0° if the glass is quite clean. Surface tension may be lowered by adding wetting agent. Corrodes iron and steel |
|
Ethyl alcohol C2H5OH |
0.8 <**> |
22 |
110 |
(**) Rises if water is present Corrodes iron and steel |
|
|
Ethylene bromide, C2H4Br3 |
2.18 |
38 |
High |
Attacks natural and synthetic rubber, corrodes iron and steel. Surface tension can be reduced by adding toluol. |
|
|
Acetylene tetrabromide C2H2Br4 |
2.98-3.00 |
Attacks natural and synthetic rubber, corrodes iron and steel. Surface tension may be reduced by adding toluol |
|||
|
Mercury Hg |
13.56 |
465 |
127° |
18 |
Combines with gold, copper and brass. Attacks iron and steel very slowly |
|
Carbon tetrachloride CCI4 |
1.58 |
26 |
0° |
124 |
Attacks rubber, but not metals |
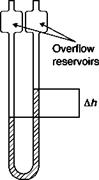 |
U-tube manometer Pi 02
pa is considered negligible with respect to pt is of the order 10-3 when water is the manometer liquid and 10-4 when mercury is used.
Usually one of the two arms of the manometer remains connected to the environment; in wind tunnels, sometimes it is connected to the static pressure of the stream, pm, so the manometer indicates the value of p – pm which actually determines the forces acting on the body: in fact, pm is the hydrostatic pressure acting all around the body, thus giving a net zero contribution.
The manometer is susceptible to blowing its liquid into the attached pressure lines if the range of the instrument is exceeded; however, all good manometers employ overflow reservoirs at the end of the tubes to retain the fluid in case of accidental overloading.
This type of manometer is very simple and requires no calibration because pressure is obtained from the analytical Equation (1.1). Furthermore, if the internal diameters of the two arms are equal, the effects of capillarity are balanced, though small errors can arise due to the fact that the tube wall next to the liquid surface is wet in the arm in which the level decreases and is dry in the other one. Errors like these can be eliminated by raising beyond the equilibrium position the column of liquid that must rise and letting it fall back into position before taking the reading.
The only adjustments necessary are those for changes from their standard values of the density of the liquid and of the length of the scale due to changes in temperature. If the level difference measured at temperature T is AhT and the scale is properly calibrated to the temperature T0, the corrected height difference, Ah, is given by:
where a[U_1] is the coefficient of linear expansion of the material of the scale and 5[U_1] is the coefficient of volumetric expansion of the manometer liquid. Under normal conditions the temperature changes are not large enough to introduce appreciable errors if the density and the calibration of the scale are referred to the standard temperature of 15°C.













